Mikro-Cath Pressure Catheter
The Mikro-Cath pressure catheter, now including a marker band, delivers repeatable, exact data for reliable insight into cardiovascular, respiratory, and intra-compartmental pressure cases. The solid-state catheter is unaffected by motion artifacts or body position, enabling data you can count on when making critical clinical decisions. The Mikro-Cath also provides expert evaluation for benchtop studies when evaluating efficacy of medical device performance.
High-Fidelity Pressure Benefits
The Mikro-Cath’s high-fidelity technology provides ease of calibration to get access to data faster, a clean, accurate signal to eliminate the guesswork during analysis, and real-time data directly from the source to assess hemodynamics, airway flow, or suspected compartment syndrome. As a gold standard method for measuring pressure, the Mikro-Cath is highly suitable for high and low pressure environments within the body. With limited noise in the signal, there is a clear understanding of the patient’s condition.
Mikro-Cath Applications
Cardiovascular Pressures1
Millar's Mikro-Cath pressure catheter delivers true hemodynamic data for deeper insight into cardiovascular function, helping guide diagnostic decisions when precise measurements count. Mikro-Cath data from the source provides reliable signals, including accurate dP/dt for advanced clinical applications, such as exercise studies, drug therapy evaluation, and heart failure cases.
Intra-Compartmental Pressures2
The Mikro-Cath™ records precise measurements within compartments of the body, typically in the upper and lower extremities, to monitor for compartment syndrome. The continuous, real-time data supports the diagnosis of acute compartment syndrome (ACS) or evaluation of chronic exertional compartment syndrome (CECS).
Airway Pressures3
Mikro-Cath Pressure Catheters use our proven high-fidelity pressure sensor technology to record precise measurements within the airway to advance discovery in respiratory physiology. Signals are unaffected by patient position or movement during respiratory cycles of breathing events such as obstructive sleep apnea.
Expert Insight into Clinical Cases
The Mikro-Cath can be easily integrated into standard patient monitoring systems via interface cables and a Millar pressure control unit, offering an immediate upgrade to the fluid-filled monitoring alternatives. There is no need to calibrate to patient height or insertion angle and the patient position does not affect the pressure signal. Continuous data can be recorded for up to 24 hours. Review the Mikro-Cath calibration video at the bottom of this page.
Mikro-Cath vs. Traditional Fluid-Filled Catheters
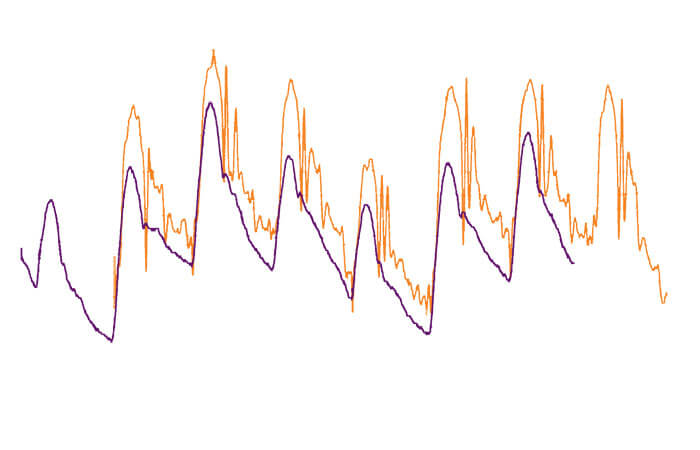
When compared to fluid-filled catheters, the Mikro-Cath Pressure Catheter provides real-time, accurate pressure data. The Mikro-Tip® sensor measures blood pressure, airway pressure, or compartment pressure at the source, providing a true representation of the pressure data and eliminating many of the common shortfalls of fluid-filled catheters, including signal variances from the patient position or timing delays. There is minimal noise and no air bubbles or motion artifacts to interfere with the signal.
Pulmonary artery pressure waveforms
- ● Fluid-filled Transducer
- ● Mikro-Cath Pressure Catheter
Specifications
| Product Reference | 825-0101 |
|---|---|
| Working Length | 120 cm |
| Tip F Size | 3.5F (1.2 mm or 0.47") |
| Body F Size | 2.3F (0.8 mm or .031") |
| Guide Catheter Compatibility | 5F |
| Tip Configuration | Straight |
| Pressure Sensor(s) | 1 |
| Use | <24 hours |
| Shaft Material | Nylon |
| Sensor Sensitivity | 5 µV/V/mmHg, nominal (37.6 µV/V/kPa) | 3.68 µV/V/cmH2O, nominal |
| Accuracy (nonlinearity, hysteresis, sensitivity and repeatability combined) | ± 1 mmHg (0.13 kPa) ±1% of reading from -50 to 50 mmHg (-6.7 to 6.7 kPa) ±3% of reading from 50 to 300 mmHg (6.7 to 40 kPa) | +/- 1.4 cmH20 +/- 1% of reading from -68 cmH20 to +68 cmH20 |
Mikro-Cath Product Downloads
Download the Mikro-Cath Pressure Catheter information sheets to learn more about Millar's market-leading solution for obtaining the most accurate and comprehensive pressure measurements.
Case Study
Advancing CECS Diagnosis in Athletes: Dr. Jaap Stomphorst’s Success with Millar’s Mikro-Cath™
Discover how Dr. Jaap Stomphorst revolutionized Chronic Exertional Compartment Syndrome (CECS) testing using Millar’s Mikro-Cath™. With over 300 patients tested, his success story showcases how real-time, continuous pressure monitoring enhances diagnostic accuracy and improves patient outcomes.
Indications for Use Statement
The Mikro-Cath Pressure Catheter is a single‐use catheter intended to be used for medical research and diagnostic purposes. The catheter is indicated to measure cardiovascular, intra-compartmental, and airway pressures in the human body. The catheter is used as a minimally invasive device under short-term limited body contact (<24 hours).
The typical cardiovascular application will be through the femoral artery with the use of an additional guiding catheter.
Mikro-Cath Pressure Catheter may be introduced into the targeted muscle compartment through an introducer. The Mikro-Cath may be introduced into the respiratory system through an existing orifice or through an incision.
Additional contraindications, precautions, and warnings are referenced in the Instructions for Use available under the Knowledge Center acute catheter manuals.
Video: Mikro-Cath Calibration
Contact Millar
Contact a Sales Rep or call Millar at +1 832.667.7000 to learn more about how our Mikro-Cath Pressure Catheter can advance your clinical research or applications.